Research Group Alessandro A. Sartori
Keywords
DNA repair pathways, Genome Instability, Synthetic Lethality, Peptide Drugs, Targeted Cancer Therapy
Summary & Mission statement
Our research aims to decipher the molecular framework of DNA damage signalling and repair for the rational design of targeted anti-cancer therapies.
Overview
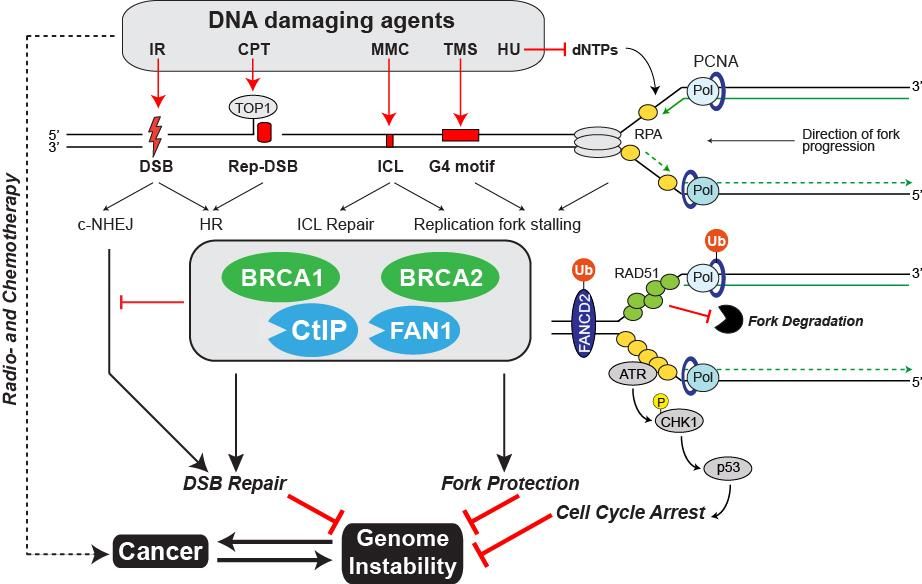
Studying the causes and consequences of genomic instability is a major topic of research within cancer biology. Paradoxically, DNA damage not only drives tumorigenesis but is also used as an effective means to kill malignant cells through radio- or chemotherapy. However, most of these treatments are non-selective causing severe side effects. Therefore, a better understanding of the underlying molecular mechanisms activated in response to DNA damage – most notably DNA repair – is of utmost importance for the discovery of novel anti-cancer drugs and the development of improved diagnosis and therapies.
The main focus of our laboratory is to gain insights into the molecular and genetic interplay between DNA repair pathways in response to different types of DNA damaging agents. Within this broad area of research, we are particularly interested in the repair of DNA double-strand breaks (DSBs) and interstrand crosslinks (ICLs), the most deleterious lesions a cell can encounter. A detailed knowledge of the players acting in DSB and ICL repair processes is imperative if we are to improve current cancer therapy.
We are employing several in vitro and in vivo approaches to investigate the function and regulation of CtIP, FAN1 and other genome caretaker proteins in response to DNA damage and replication stress. For instance, CtIP and FAN1 activities are governed by multiple protein-protein interactions (PPIs). Consequently, selective targeting of CtIP or FAN1 PPI interfaces using synthetic peptides in combination with DNA damaging agents is expected to act synergistically in eliminating cancer cells.