Research Group Stephanie Tanadini-Lang
Our mission is to integrate advanced technologies into clinical practice while rigorously evaluating their potential benefits and risks for patients. Our group has pioneered the implementation of cutting-edge technologies such as Artificial Intelligence (AI), MR Linac, and MR-only workflows. We are also at the forefront of research in imaging biomarker detection, Flash radiotherapy, and AI developments. Explore our site to learn more about our innovative projects and how we are shaping the future of medical physics.
Quantitative Image Analysis
Cancer is a heterogeneous disease, varying in etiology, pathogenesis, therapy response, and prognosis. Tumor response to treatment differs not only among patients but also within individual tumors. With rapid technological advancements, an increasing number of cancer treatment options are available. Therefore, decision support systems are essential to provide the right treatment for each patient.
One way to optimize treatment strategies is through the identification of biomarkers. Recently, imaging has gained importance for identifying new prognostic biomarkers due to its non-invasive nature. Imaging datasets contain more information than what is visible to the human eye. Radiomics involves extracting a large number of meaningful quantitative features from medical images, such as computed tomography (CT), positron emission tomography (PET), or magnetic resonance imaging (MRI).
Using dedicated software we can extract radiomic features that describe tumor shape, intensity, and texture from medical images. Based on mathematical definitions, we investigate tumor morphology and prominent perceptual texture characteristics such as regularity (periodicity), directionality, and complexity. These radiomic features are potential biomarkers of the cancer phenotype and can be used for patient outcome prognosis or correlated to tumor biology using advanced statistical methods.
Related Projects
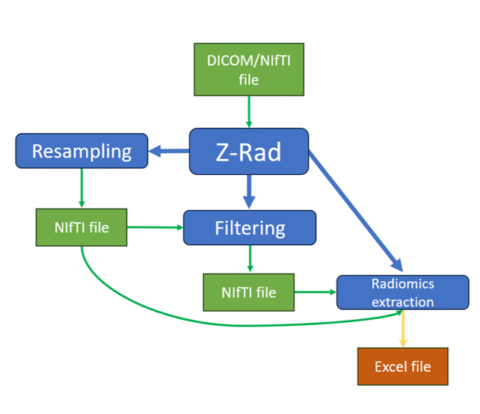
Z-Rad is a user-friendly radiomics extraction software written in Python that features a graphical user interface (GUI) and an application programming interface (API).
Z-Rad supports the most common clinical imaging formats, DICOM and NIfTI, and processes different imaging modalities using specific methods. For DICOM data, CT images are automatically converted to Hounsfield Units (HU) based on DICOM attributes. For PET images, the Standardized Uptake Value (SUV) is calculated using the body weight approach, taking into account vendor-specific DICOM attributes. MR images are read directly from the DICOM array without preprocessing.
The GUI is divided into three tabs, each tailored for specific operations:
- Resampling: This tab supports image resampling alone, alongside regions of interest (ROI) masks, or converting DICOM files to NIfTI images and masks without resampling. Resampling can be performed in 3D or 2D (axial slice-wise) using nearest neighbor, linear, B-spline, and Gaussian strategies.
- Filtering: Users define the desired filter settings. Z-Rad supports mean, Laplace of Gaussian, Laws kernels, and wavelet (Daubechies 2, Daubechies 3, first-order Coiflet, and Haar) filters. Detailed filtering information is available in the user guide on the Z-Rad website.
- Radiomics: Parameters for radiomics feature extraction include intensity re-segmentation (e.g., HU for CT or SUV for PET within ROIs), intensity outlier filtering, discretization strategies, and various radiomics feature aggregation methods covering 2D, 2.5D, and 3D options. Radiomic features include shape, intensity, grey level co-occurrence matrix (GLCM), grey level run length matrix (GLRLM), grey level distance zone matrix (GLDZM), neighboring gray tone difference matrix (NGTDM), and neighboring gray level dependence matrix (NGLDM) families.
The Z-Rad API allows seamless integration into any Python-based research workflow, offering rich functionalities for resampling, filtering, and radiomics.
PIs:
- Stephanie Tanadini-Lang
Researchers:
- Maksym Fristak
- Hubert Gabrys
Funding: The CCCZ oligometastatic disease program – OMD
Aim (short background and aims of the project) Early personalized health initiatives and projects have uni-dimensionally focused on biological sample-based disease phenotyping. However, environmental factors are known to attenuate the association between the patients` genetic background and outcome. Consequently, the need for analysis of “downstream” phenotypes has been recognized and has become a strong field of research within personalized health. In this context, multi-modal medical imaging is an important source of phenotype information and plays a prominent role in the diagnosis, staging, and treatment response monitoring. Despite the fact that imaging is standard practice to characterize the patient’s individual disease and to estimate patient individual outcome, the incorporation of medical images into personalized health has been blocked by the current standard practice of image analysis: image analysis is mostly a manual process resulting in predominantly qualitative image characterization. To unlock the full potential of medical images for personalized health, quantitative image characterization (Radiomics) has become a highly promising field of research.
In this driver project, we propose an initiative to build a Swiss-wide infrastructure for image-based biomarker research & analysis. We will standardize imaging, image analysis and image-based outcome modeling to evaluate the value of MR images acquired in clinical routine as prognostic and predictive biomarkers in patients treated for Glioblastoma multiforme. We hypothesize that radiomic biomarkers will improve outcome modeling and improve selection of the most appropriate patient-individual treatment strategy.
PIs:
- Matthias Guckenberger, USZ
Researchers:
- Bram Stieltjes, University Hospital Basel
- Maricio Reyes, University of Bern
- Roland Wiest, University Hospital Bern
- Christoph Stippich, University Hospital Zürich
- Nicolaus Andratschke, University Hospital Zürich
- Reto Meuli, CHUV Lausanne
- Cornelia Kruschel Weber, University Hospital Zürich
- Stephanie Tanadini-Lang, University Hospital Zürich
- Adrien Depeursinge, HES-SO Valais
- Olivier Michielin, CHUV Lausanne
- Giorgio Treglia, Ente Ospedaliero Cantonale
Funding: This project is supported by the Swiss Personalised Health Network (SPHN): 2019 – 2021
Publications:
- https://doi.org/10.1007/978-3-031-09002-8_33
- https://doi.org/10.1016/j.phro.2022.05.006
- https://doi.org/10.1148/radiol.231319
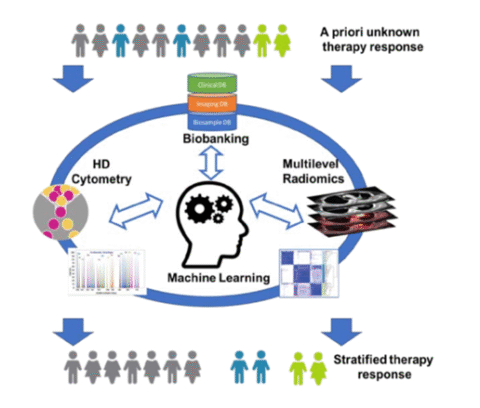
Immunotherapy has revolutionized cancer treatment and renewed efforts to find therapeutic targets that enhance anti-tumor immunity across all types of malignancies. Checkpoint inhibition (CPI), CAR-T cells, and cytokines are now combined with conventional approaches to treat cancer and stimulate anti-tumor immunity. Clinical trials often measure success through general clinical parameters, with outcomes based on radiological imaging and survival rates. However, this conventional monitoring has a downside: patient responses are often detected long after treatment initiation, causing therapy-resistant patients to experience adverse effects and delaying potentially better-tailored therapies. Predictive and monitoring biomarkers for immunotherapy are just beginning to emerge, while new therapeutic approaches still largely depend on conventional clinical monitoring.
Although biased analysis of blood parameters, targeted cytometry, and bulk-omics have provided some insights into potential biomarkers, high-dimensional single-cell cytometry combined with unbiased algorithm-based pattern recognition has revolutionized biomarker identification and personalized medicine. Similarly, advances in radiological imaging, particularly radiomics, now allow for more precise determination of tumor burden, cancer characterization, and assessment of treatment response.
This project focuses on malignant melanoma patients undergoing immune checkpoint inhibition (aPD-1 or aPD-1/aCTLA-4). We aim to:
- Identify biomarkers to predict the therapeutic response to combination cancer immunotherapy.
- Monitor therapy responses during combination therapy.
- Analyze polymorphonuclear granulocytes as indicators of anti-tumor immunity.
The project combines cutting-edge clinical oncology, computational radiological image analysis, and innovative computational biology to identify prognostic and predictive signatures. Our goal is to establish clinically applicable tools to guide precision medicine decisions and identify new therapeutic targets for cancer treatment.
PIs:
- Burkhard Becher, Institut für Experimentelle Immunologie, UZH
- Manfred Claassen, Institut für Molekulare Systembiologie ETH Zürich
- Matthias Guckenberger, Department for Radiation Oncology, USZ
- Mitchell Levesque, Department of Dermatology, USZ
Researchers:
- Hubert Gabrys, Department for Radiation Oncology, USZ
Funding: Schweizerischer Nationalfonds (SNF) Sinergia: 2019 – 2022
Publications:
Project Title Radiomics analysis for differentiating between oligometastatic and polymetastatic disease, regardless of the primary cancer type
Aim (short background and aims of the project) Oligometastatic cancer is characterized by a limited number of metastases, typically restricted to one or a few organs, representing an intermediate stage between localized and widely metastatic cancer. Despite its clinical significance, distinguishing oligometastatic from more advanced metastatic cancer is challenging due to the lack of clear biological markers and standardized diagnostic criteria, complicating tailored treatment strategies. Therefore, there is a strong rationale for exploring automated tumor lesion detection and radiomics.
This project aims to identify prognostic and predictive imaging biomarkers to distinguish between oligometastatic and polymetastatic disease. By combining expertise in molecular biomarkers, quantitative radiomic research, and machine learning, we will analyze the heterogeneous growth patterns of these cancers. For patients with various primary tumors, we aim to conduct automated longitudinal segmentation of all tumor lesions and analyze radiomic signatures to differentiate between oligometastatic and polymetastatic disease.
PI:
- Matthias Guckenberger
- Stephanie Tanadini-Lang
Researchers:
- Maksym Fritsak
Funding: The CCCZ oligometastatic disease program – OMD
Treatment with immune-checkpoint inhibitors has significantly improved outcomes in metastatic non-small cell lung cancer (NSCLC), making it a first-line treatment. However, only up to 50% of patients respond to immunotherapy, and many experience disease recurrence. As a result, new treatment strategies are being investigated. Recent randomized studies have demonstrated a survival benefit when local consolidative therapy is added to standard systemic therapy for patients with metastatic NSCLC. These studies involved the local eradication of all cancer lesions in addition to systemic therapy and were limited to patients with low metastatic tumor burden, known as oligometastatic disease.
Oligometastatic disease is defined by the presence of a small number of radiologically visible metastases. However, the number of metastases is an imprecise surrogate marker for limited metastatic capacity, highlighting the need for novel biomarkers to accurately identify oligometastatic patients and distinguish them from those with more advanced, polymetastatic disease.
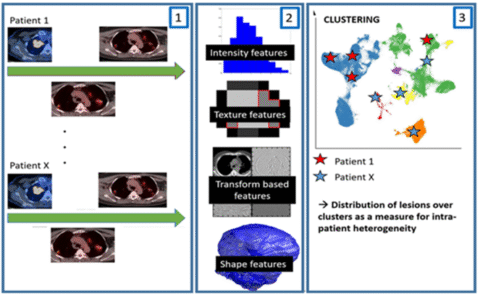
Radiomics has demonstrated potential in identifying novel prognostic imaging-based biomarkers in various cancer types, including NSCLC treated with immunotherapy. Our research group has shown the prognostic value of CT- and FDG-PET-based radiomics in radiotherapy for head and neck cancer, demonstrating the added value of radiomic biomarkers compared to HPV status. Radiomics is particularly promising for metastatic disease as it allows longitudinal individual analysis of each metastatic lesion.
In this project, our aim is to use radiomics to identify patients with truly oligometastatic lung cancer and distinguish them from those with polymetastatic disease. With FDG-PET/CT imaging available at multiple time points and our in-house developed radiomics software, we will characterize and compare different tumor foci (primary tumor, lymph node metastases, distant metastases) through cluster analysis. This approach enables us to investigate how individual tumor foci change over time. We will test the prognostic relevance of this tumor heterogeneity and determine whether it allows differentiation between oligometastatic and polymetastatic tumors.
PIs:
- Matthias Guckenberger, Department for Radiation Oncology, USZ
- Stephanie Tanadini-Lang, Department for Radiation Oncology, USZ
Researchers:
- Alina Paunoiu, Department for Radiation Oncology, USZ
Funding: Foundation for Applied Cancer Research
Artificial intelligence in radiation oncology
Artificial intelligence (AI) in radiation oncology potentially offers significant advancements by automating routine tasks, which can optimize time allocated to standard cases and allows clinical staff to focus on more complex and critical cases. One notable example is the use of synthetic CT, which eliminates the need for separate CT simulation for the patient and enables a MR-only workflow for radiotherapy treatment planning. The latter provides advantages in terms of uncertainties in image registration, radiation exposure to the patient and workflow. However, integrating AI solutions into clinical practice introduces new challenges in quality assurance, necessitating robust validation and continuous monitoring to ensure safety and efficacy in patient care. These topics are addressed by our research group, which aims to develop in-house algorithms for radiotherapy planning automation, synthetic CT generation and quality assurance.
Synthetic CT (sCT) imaging has emerged as an innovation in radiotherapy planning, offering a promising alternative to simulation CT scans. The generation of synthetic CT images can be performed using neural networks, which have shown superiority compared to previously proposed methods. The general aim is predicting the electron density information required for accurate dose calculations and patient positioning, which are traditionally based on a simulation CT scan. Our group develops neural networks that are trained on extensive datasets, learning to convert MR images into synthetic CT images. Treatments sides such as brain and pelvis have been extensively researched in the last few years and commercial solutions are available for clinical implementation. Further body sides such has the abdomen and the head-neck region still require further developments. Our group focused on the latter, developing and proposing novel algorithms for sCT generation. The promising results have shown applicability of sCT in these body sites. The accuracy and precision of synthetic CT images are critical for their implementation into clinical practice. High fidelity in sCT imaging ensures that dose calculations are as precise as those based on conventional CT scans. Normally, a dose consistency to the 2% level is required, while 1% could be achieved with high quality sCT. Therefore, the neural networks used in sCT generation must be rigorously trained and validated to meet these clinical standards. Moreover, precision in sCT imaging is vital for daily patient positioning. These synthetic images are compared to the CBCT or planar x-rays acquired while the patient is on the treatment couch. A precise alignment of the patient is crucial for targeting tumors and sparing normal tissue. The required level of patient position accuracy depends on the treatment technique and can reach the sum-millimeter level for brain stereotactic treatments. Our research groups evaluate and develop validation approaches for patient positioning based on synthetic images. An example is a novel approach based on contours to CBCT matching. In such a case, the contours delineated on the MR are matched directly to the daily CBCT image, which can then be retrospectively evaluated in terms of MR-CBCT or sCT-CBCT match quality. Finally, further advantages of radiotherapy planning based on sCT include: reduced radiation exposure for the patient, reduction of the hospital visits for the patient, simplification of the workflow for the staff and reduction of the uncertainties in the image to image matching. Our research group works on the developments necessary to bring such potential advantages from the research field into clinical practice.
Researchers / PIs:
- Mariia Lapaeva
- Francesca Nella
- Riccardo Dal Bello
Funding:
- USZ Innovationspool 2023
- Siemens Healthineers collaboration
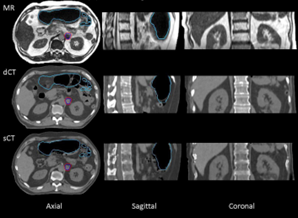
Example of synthetic CT generation in the abdominal region with an in-house developed software. Figure from paper: https://doi.org/10.1016/j.phro.2022.11.011
Original research article: https://doi.org/10.1016/j.phro.2022.11.011
Original research article: https://doi.org/10.1016/j.phro.2023.100464
Original research article: https://doi.org/10.1016/j.phro.2023.100471
Review article: https://doi.org/10.1016/j.radonc.2024.110387
White paperSynthetic CT (sCT) imaging has emerged as an innovation in radiotherapy planning, offering a promising alternative to simulation CT scans. The generation of synthetic CT images can be performed using neural networks, which have shown superiority compared to previously proposed methods. The general aim is predicting the electron density information required for accurate dose calculations and patient positioning, which are traditionally based on a simulation CT scan. Our group develops neural networks that are trained on extensive datasets, learning to convert MR images into synthetic CT images. Treatments sides such as brain and pelvis have been extensively researched in the last few years and commercial solutions are available for clinical implementation. Further body sides such has the abdomen and the head-neck region still require further developments. Our group focused on the latter, developing and proposing novel algorithms for sCT generation. The promising results have shown applicability of sCT in these body sites. The accuracy and precision of synthetic CT images are critical for their implementation into clinical practice. High fidelity in sCT imaging ensures that dose calculations are as precise as those based on conventional CT scans. Normally, a dose consistency to the 2% level is required, while 1% could be achieved with high quality sCT. Therefore, the neural networks used in sCT generation must be rigorously trained and validated to meet these clinical standards. Moreover, precision in sCT imaging is vital for daily patient positioning. These synthetic images are compared to the CBCT or planar x-rays acquired while the patient is on the treatment couch. A precise alignment of the patient is crucial for targeting tumors and sparing normal tissue. The required level of patient position accuracy depends on the treatment technique and can reach the sum-millimeter level for brain stereotactic treatments. Our research groups evaluate and develop validation approaches for patient positioning based on synthetic images. An example is a novel approach based on contours to CBCT matching. In such a case, the contours delineated on the MR are matched directly to the daily CBCT image, which can then be retrospectively evaluated in terms of MR-CBCT or sCT-CBCT match quality. Finally, further advantages of radiotherapy planning based on sCT include: reduced radiation exposure for the patient, reduction of the hospital visits for the patient, simplification of the workflow for the staff and reduction of the uncertainties in the image to image matching. Our research group works on the developments necessary to bring such potential advantages from the research field into clinical practice.
Researchers / PIs: Klara Kefer, Stefanie Ehrbar, Riccardo Dal Bello
Funding: SASRO Research Grant 2022
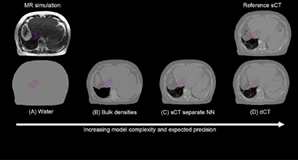
Research output:
Original research article:
https://doi.org/10.1016/j.zemedi.2023.02.003
Original research article:
https://doi.org/10.1016/j.phro.2023.100464
Original research article:
https://doi.org/10.1016/j.phro.2020.09.013
Original research article:
https://doi.org/10.1016/j.ctro.2023.100624
Brain metastases are among the most common neurological disorders, particularly in patients with lung and melanoma cancers. According to the literature, 40% of patients develop more than three metastases. Contrast-enhanced MRI is the key imaging modality for diagnosing and following up with patients who have brain metastases. Precise detection and delineation of brain metastases on MRI scans are crucial in radiation oncology for treatment planning and assessing response to therapy over time. However, manual segmentation is tedious, time-consuming, and prone to inter-observer variability.
Our group is developing and implementing a deep learning-based method for auto-contouring brain metastases. Despite the promising results of deep learning methods in auto-detection and segmentation, concerns remain regarding their implementation in daily clinical workflows due to safety issues and the overconfident predictions of deep learning models, even when they are incorrect.
Our research focuses on quantifying the uncertainty related to both the model and the data in brain metastases segmentation. By estimating uncertainty, we can enhance the safety of auto-segmentation by flagging uncertain cases for physician review and correction. This approach ensures more accurate and reliable segmentation, facilitating the safe integration of deep learning methods into clinical practice.
PIs:
- Stephanie Tanadini-Lang
- Nicolaus Andratschke
Researcher:
- Zahra Khodabakhshi
Funding:
CRPP- Clinical Research Priority Program Grant on “Artificial Intelligence in Oncological Imaging” University of Zurich
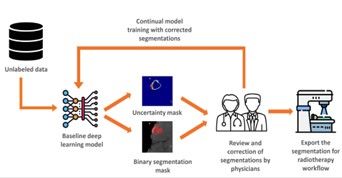
Research output:
FLASH Radiotherapy
FLASH radiotherapy is an experimental technique aimed at maintaining tumor control while reducing normal tissue toxicity. This could potentially expand the therapeutic window. This sparing effect has been demonstrated in preclinical models and can be achieved when radiotherapy is applied at ultra-high dose rates (>40 Gy/s, compared to about 6 Gy/min in conventional therapy). We are currently planning a clinical Phase I study for the treatment of skin metastasis of melanoma. Only a few accelerators worldwide can generate ultra-high dose rate beams, and we are among the first centers to conduct a clinical study using this innovative technique. Our research group works on the technical preparation for the clinical study and in parallel at pre-clinical biology experiments. The challenges of this research topic involve the use of ionizing radiation at dose rates far beyond the range for which conventional detectors are designed for and the absence of redundant feedback systems within the accelerator. Our research group works on characterizing novel detectors for FLASH radiotherapy, developing quality assurance programs for ultra-high dose rate electron accelerators and investigating the FLASH effect in pre-clinical models for deciphering the underlying mechanism.
FLASH radiotherapy with electron beams represents a potential advancement in the field of cancer treatment. This technique uses ultra-high dose rates to potentially spare normal tissue while maintaining tumor control. Our research group implemented this innovative approach by modifying a decommissioned linear accelerator, which had been in clinical use from 2009 until 2022. We demonstrated that dose rates above 1 Gy per pulse and 200 Gy per second at the isocenter can be achieved for 16 MeV and 9 MeV electron beams. We plan to employ this modified linac for a phase I feasibility and safety clinical trial. Current research work focuses on the technical preparation for the clinical trial. One of the primary goals of current research is to assess the stability and reproducibility of the beam properties on the short and long term. Achieving consistent beam characteristics is crucial for ensuring that FLASH radiotherapy can be reliably used in clinical settings. This involves detailed evaluations of the beam’s output, energy, and profiles. To address these challenges, we are developing new methods to perform quality assurance (QA) of the FLASH beam. Traditional QA techniques used in conventional radiotherapy are not always directly applicable to the ultra-high dose rates characteristic of FLASH radiotherapy. Therefore, innovative approaches are required to ensure the accurate delivery of such high doses. One example is the characterisation of prototype active detectors suitable for ultra-high dose rates and their use in in-house developed phantoms for end-to-end testing. The end-to-end testing are performed to simulate clinical conditions and verify the entire treatment process, from planning to delivery. Moreover, we investigate the use of passive detector in collaboration with the Paul Scherrer Institute (PSI). Also, beam stability and reproducibility over time are critical topics in this project. Long-term studies are being conducted to monitor the performance of the modified linac, identifying any trends or deviations that could impact its clinical use. This involves continuous data collection and analysis, allowing the maintenance of optimal performances. Within this project we modified a decommissioned linac for investigating FLASH radiotherapy with electron beams. Current work focuses on the assessment of the stability and reproducibility of the beam, aiming to use FLASH radiotherapy within a clinical trial.
Researchers: Jens von der Grün, Serena Psoroulas, Dominik Flückiger, Riccardo Dal Bello
Funding: Varian medical systems collaboration
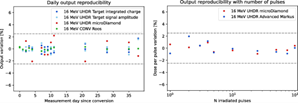
Example of assessment of the linac stability for the delivery of ultra-high dose rate beams. Figure from article: https://doi.org/10.1016/j.radonc.2023.109822
Research output:
Original research article:
https://doi.org/10.1016/j.radonc.2023.109822
Original research article:
DOI 10.1088/1361-6560/ad1cf5
Original research article:
https://doi.org/10.1016/j.radmeas.2024.107227
The delivery of radiotherapy (RT) with ultra-high dose rates (UHDR) offers a promising opportunity to widen the therapeutic window. This is potentially achieved by an increased sparing of normal tissue while maintaining unchanged tumor control, a phenomenon known as the FLASH effect. The field of research is evolving rapidly, with the first in-human applications of electron and proton UHDR paving the way for further clinical trials focusing on patients with melanoma and bone metastasis. These clinical trials are supported by numerous positive experiments demonstrating superior normal tissue sparing in various animal models, including rodents, zebrafish, mini-pigs, and domestic animals. However, some experiments have reported negative results, particularly with rodents, zebrafish, and domestic animals. Understanding the presence or absence of the FLASH effect could provide insights into the underlying mechanisms, with oxygen content and UHDR beam properties playing crucial roles. These properties can be explored with experimental irradiation platforms that offer a wide range of beam parameters. Extending these approaches to even more diverse beam configurations is a key aim of this work. Previous in-vivo studies have relied on animal models that must adhere to strict ethical regulations, limiting the scalability and throughput of experiments. To address this, we are focusing on applying this novel irradiation technique to in-vivo experiments using animal models with looser ethical requirements, allowing for higher throughput. One such model is Drosophila melanogaster, which has been widely used in radiation research but has not been extensively studied in the context of UHDR RT and the FLASH effect. Using Drosophila melanogaster as a model organism offers several technical benefits compared to vertebrate models. These include simplicity and cost-effectiveness in laboratory maintenance, a significantly shorter life cycle, abundant externally laid embryos, and the ability to undergo various genetic modifications. However, a notable drawback is the higher radioresistance of Drosophila melanogaster compared to vertebrate animals. Reducing the lifespan of adult flies requires hundreds to thousands of Grays, while observing endpoints in larvae necessitates tens to hundreds of Grays. Despite this, Drosophila melanogaster presents a promising and novel model for investigating UHDR RT and can significantly increase research throughput. Our research group, in collaboration with the biology research group, aims to investigate the FLASH effect in Drosophila melanogaster. This includes the development of dedicated irradiation setups, their characterisation and the animals follow-up post irradiation.
Researchers / PIs: Marvin Kreuzer (UZH – Biology), Irene Vertugno (UZH – Biology), Martin Pruschy (UZH – Biology), Riccardo Dal Bello

Overview of the experimental setups developed and characterized for delivering radiation to Drosophila melanogaster. Figure from abstract: https://user-swndwmf.cld.bz/ESTRO-2024-Abstract-Book/3330/
Research output / publications:
https://user-swndwmf.cld.bz/ESTRO-2024-Abstract-Book/3330/


