Translational Diabetes
We investigate how external influences and environmental factors - such as a high-fat diet or air pollution - can affect the immune system in the gut and thereby alter our health, particularly our metabolism (e.g. body weight and diabetes risk).
AG Cavelti-Weder / Translational Diabetes
The exposome describes the totality of all environmental factors to which a person is exposed in the course of their life. These include exogenous influences such as the food we eat or air pollutants we breathe in, but also endogenous factors such as the microbiome in the gut. These environmental factors interact with our genetic predisposition and have a significant influence on our health.
In our research group, we investigate selected components of the exposome and their influence on the intestinal immune system. We are particularly interested in how nutrition, pollutant particles, plasticizers or infectious agents can trigger inflammatory processes in the intestine via the immune system and thus contribute to the development of widespread metabolic diseases such as obesity and diabetes.
Using in vivo mouse models and translational studies in humans, we analyze the changes in the innate and adaptive immune system in the gut in relation to obesity and diabetes. Furthermore, we are interested in the underlying mechanisms of these disease processes and whether the associated immunological changes in the gut are reversible by weight reduction.
These findings provide new insights into the immune functions of the gut and form the basis for targeted interventions against environmentally induced metabolic diseases.
Research topics
Basic research I: Role of nutrition in intestinal (patho)physiology and metabolic consequences
Metabolic diseases such as obesity and diabetes are increasingly recognized as complex diseases that are triggered by genetic, nutritional and environmental factors. Nutrition plays a crucial role in modulating mucosal immunity with effects on glucose homeostasis and metabolic health. We have shown that a high-fat diet leads to an increase in inflammatory macrophage populations in the intestines of mice [1]. We have transferred these results to humans and confirmed that obesity and an unhealthy lifestyle alter immune cell populations in the human gut [2]. Specific depletion of macrophages in the large intestine of mice fed a high-fat diet resulted in improved glucose tolerance, confirming a causal link between macrophages in the large intestine and glucose homeostasis [1]. However, a long-term comprehensive analysis of intestinal immunity in obesity, its potential reversibility and the mechanistic relationships is still poorly understood.
We want to understand how a high-fat diet affects immune cell populations in the gut and how these changes contribute to metabolic disorders. The ultimate goal is to translate mechanistic insights into new therapeutic strategies to improve metabolic health and prevent diabetes.
Specific projects:
- Dynamic changes in intestinal immune cells in obesity over time
- Effects of dietary fats on glucose homeostasis and immune cells
- Effects of weight loss on immune cell populations
- Mechanism of the interferon response induced by a high-fat diet
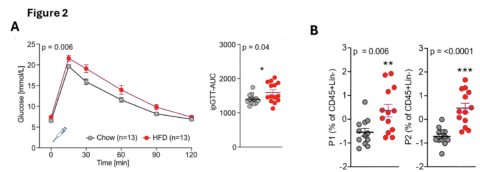
Figure 2: Glucose intolerance and inflammatory macrophages in mice fed a high-fat diet compared to a standard diet.(A) Glucose tolerance test of mice after one week on a high-fat diet (red) compared to mice on a standard diet (gray).(B) Z-score of the frequency of proinflammatory macrophage populations P1 and P2, normalized to CD45+ cells.
Basic research II: Role of environmental factors in intestinal (patho)physiology and metabolic consequences
In addition to the generally known risk factors for diabetes such as diet, lifestyle and genetics, several environmental factors are known to increase the risk of diabetes. For example, air pollution and smoking have been identified as risk factors for diabetes in both epidemiologic studies [3-6] and rodent studies [7, 8]. Traditionally, the lungs have been considered the organ primarily affected by these pollutants. However, the role of the gastrointestinal tract has been largely overlooked, despite evidence that these particles enter the gut via mucociliary clearance from the lungs and by direct ingestion via contaminated food. To determine the downstream metabolic consequences of lung and gut exposure to air pollution, we exposed mice to air pollution particles directly in the lung (intratracheal instillation) or in the gut (special diet). We found that oral, but not intratracheal, exposure to air pollution particles induced an inflammatory environment in the gut with subsequent beta cell dysfunction [9]. Mechanistically, oral exposure to air pollution particles led to a reduction in anti-inflammatory/resident macrophages and an interferon and inflammatory signature in colonic macrophages [10]. Depletion of macrophages, NLRP3 or IL-1β protected mice from air pollution-induced glucose intolerance, demonstrating their involvement in the pathogenesis of the disease [10].
Several environmental factors have been associated with an increased risk of diabetes. Our aim is to investigate whether the mechanisms involved are similar to those of air pollution particles, namely by inducing an inflammatory milieu in the gut.
Specific projects:
- Oral exposure to cigarette smoke particles and metabolic dysregulation
- The role of plasticizers and substitute products in metabolic diseases
- The intestinal protozoa Trichomonas musculus and glucose homeostasis
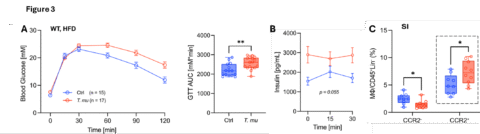
The T. mucolonization in wild-type mice fed a high-fat diet leads to impaired glycemic control, increased insulin secretion and intestinal inflammation. (A) Intraperitoneal glucose tolerance test (ipGTT), area under the curve (AUC) of T. mu-colonized (red) and non-colonized (blue) mice. (B) Circulating insulin measured during the first 30 minutes of ipGTT. (C) Inflammatory CCR2+ macrophages in distal small intestinal tissue.
Translational studies on the basis of basic research
Our overarching goal is to bridge the gap between preclinical research and translational applications by validating results in human studies. An example of this approach is our work on mucosal immunity in mice on a high-fat diet [1], which we have successfully translated to humans with obesity [2]. We have also established two mouse models for weight loss surgery and demonstrated that a long biliopancreatic arm is metabolically superior to a short arm [11, 12]. To translate these findings into a clinical context, we are investigating the metabolic and immunologic effects of a short versus a long biliopancreatic limb in human patients undergoing bariatric surgery. In addition, we are using RNA sequencing data from peripheral blood mononuclear cells of patients undergoing weight loss surgery to identify early biomarkers of metabolic improvement. In parallel, we are analyzing an existing dataset on the gut microbiome to assess the translational relevance of our preclinical findings on the role of Trichomonas species in glucose metabolism.
We want to understand the reversibility of changes in immune cells after weight loss surgery and the relationship between Trichomonas species and the risk of diabetes in humans.
Specific projects:
- Effects of weight loss surgery on intestinal immune cells(SLIM study)
- Effects of weight loss surgery on the blood transcriptome
- Trichomonas and glucose homeostasis in humans
Clinical Trials
As a senior physician in the Department of Endocrinology and Clinical Nutrition at the University Hospital Zurich, I lead several studies focusing on inpatient and outpatient diabetes care. The research questions we address are directly derived from clinical challenges and unmet needs and aim to improve patient management, optimize therapeutic strategies and integrate innovative technologies into diabetes care. An important part of these efforts is the national diabetes registry SwissDiab, in which I am actively involved as a board member. This registry is a valuable source of longitudinal data on diabetes patients treated as outpatients.
Concrete projects
- SwissDiab register: patient results
- Patient-reported outcomes such as quality of life in relation to diabetes complications,
- The use of finerenone in type 2 diabetes,
- Off-label use of GLP-1 receptor agonists in type 1 diabetes,
- Effect of GLP-1 receptor agonists in metabolic steatotic liver disease (MASLD).
- Continuous glucose monitoring data for advanced glycemic assessment
- C-peptide/glucose ratio for the early detection of diabetes in cystic fibrosis
- Personalized insulin dosing through machine learning
- Octopatch: A buccal patch for the administration of peptide drugs
Cooperations
- M. Rothenbühler, A. Lizoain (DCB Bern)
- D. Klein Cerrejon and Dr. N. Paunovic (ETH/ OBaris)
- M. Brändle, HOCH
- A. Odermatt (University of Basel)
- P. Vonaesch (University of Lausanne)
- S. Erttmann (Kiel University)
- N. Perakakis (TU Dresden)
- R. Peterli and Dr. R. Schneiter (Visceral Surgery, University of Basel)
- M. Scharl and Prof. L. Biedermann (Gastroenterology, USZ);
- N. Bukolich (ETH ZH)
- P. Schär (University of Basel)
- J. Roux (University of Basel)
- K.-H. Herzig (University of Oulu)
- M.-R. Jarvelin (Kings College London)
- A. Blakemore (Brunel University)
- M. Krauthammer (University of Zurich/ USZ)
Fundings
- SCAHT research program 2025-2028, Co-PI
- International Research Training Group (IRTG) between the Technical University of Dresden, the University of Zurich and ETH Zurich
- USZ innovation pool
- SNSF, project funding (320030_219851)
- SNSF, project grant (32003B_204937), Co-PI
- Vontobel-Stiftung

