Aortic aneurysm and rupture risk stratification
The aim of our research is to explore and better understand the mechanisms and processes leading to AA rupture in order to create a scientific basis for the development of novel diagnostic and therapeutic strategies.
Aortic aneurysm (AA) is defined as an enlargement of the aorta exceeding 50% of that of normal diameter. Aneurysm of the aorta is most common with >85% in the region of abdomen and is called abdominal aortic aneurysm (AAA). The prevalence of AAA with 3.0 cm is 4 – 8% for males over 65 years and 0.5 – 1.5 for females [Haertl 2020]. In Germany, 284’000 – 568’000 males over 65 years are affected. Due to the fact that the risk of rupture increases significantly at >5.0 cm, estimate number of risk patients in Germany is about 45’000. Furthermore, because aneurysm is mostly asymptomatic, it goes undetected until rupture, resulting in a mortality rate of >80%.
Currently, the aortic diameter >5.5 cm and its rapid progression of >0.5 cm within 6 months are the only reliable determinants of the imminent rupture. Consequently, patients exceeding 5.5 cm generally undergo surgical or endovascular intervention. Thereby, smaller aneurysms can rupture as well and larger aneurysms can remain stable. Thus, reliable diagnostic tools are necessary to recognize patients at increased risk of rupture. Besides, many of the molecular-biological processes leading to rupture are still unexplained.
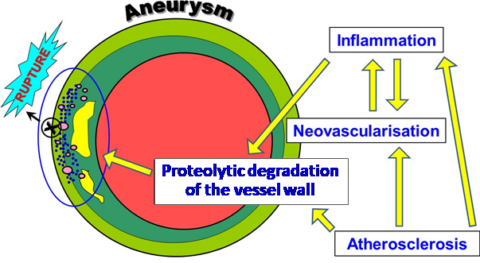
In addition, aortic dissection, even if rare, is a highly life-threatening condition, with a lethality of 1-2% every hour after the onset of the symptoms. Causing devastating complications, aortic dissection leads to death in 26% even if the patients receive a surgical intervention [Gawinecka 2017]. Without surgery, up to 58% if they are treated non-invasively. The progressive separation of the aortic wall layers results in formation of a false lumen, which can be followed by rupture or re-entry back into the true lumen. The false lumen enlarges by thrombus formation and compresses the true lumen, leading to life-threatening condition. Aortic dissection is the result of ill changes within the vessel wall by inflammatory processes, atherosclerosis and aneurysm leading to the overexpression of various proteolytic enzymes degrading in particular collagen. elastin and proteoglycans and finally to the rupture and death of the affected person.
The aim of our research is to explore and better understand the mechanisms and processes leading to AA rupture in order to create a scientific basis for the development of novel diagnostic and therapeutic strategies. For these purposes, we perform various deep analyses of ruptured and elective AAs using transcriptomics, proteomics and scRNA-sequencing focusing in particular on proteolytic enzymes (MMPs, cathepsins, ADAMs, ADAMTS) and structural proteins of the vessel wall (collagen I and III, elastin, proteoglycans). In addition, we are also interested in epigenetic changes and expression of non-coding RNAs (lncRNAs, circRNAs) in the diseased aorta.
In particular, we are also interested in molecular mechanisms that lead to aortic dissection, because its appearance progressively increases with the aortic diameter. Thus we strive to compare composition of the aortic wall in AA patients with and without dissection.
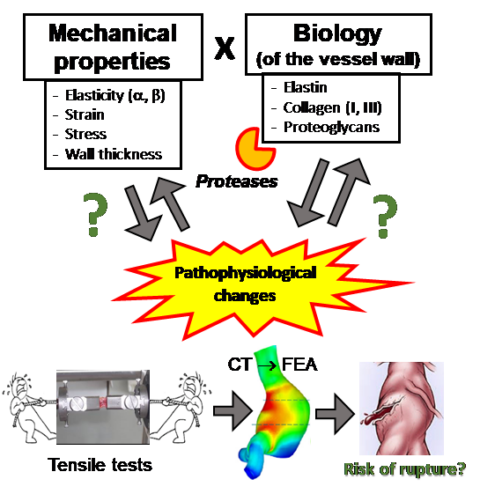
It is also to emphasise that the enlargement and rupture of the aorta is subjected to the physical laws of mechanics. Therefore, the knowledge of mechanical properties of the diseased aortic wall is of enormous importance to understand and properly predict the potential risk of rupture. The most important components of the vessel wall contributing to its stability are proteins of the extracellular matrix such as collagens, elastin and proteoglycans.
Consequently, we have been focusing also on the mechanical properties of AAA, in cooperation with the Department of Mechanical Engineering (TU Munich, Prof. Dr.-Ing. Michael Gee), performing tensile tests and finite element analyses (FEA) [Bieler 2017, Reeps, 2014, Reeps 2013, Reeps 2009]. Our results revealed that in particular collagen I and re responsible for the stability and stiffness of the aorta. Elastin fibres are mostly degraded in advanced aneurysms and do not play a relevant role anymore. In contrast, dermatan and chondroitin sulphate are responsible for aortic wall elasticity, in particular in the end stage, before the aortic wall ruptures. FEA results confirmed that the aneurysm form and shape also significantly contribute to the risk of rupture.
- He Y, Xing J, Wang S, Pelisek J, Jiang H, Xin S, Han Y, Zhang J. Increased m6A mRNA methylation levels are correlated with the progression of human abdominal aortic aneurysm. Epigenetics 2020; submitted.
- Li DY, Busch A, Jin H, Chernogubova E, Pelisek J, Karlsson J, Sennblad B, Liu S, Lao S, Hofmann P, Bäcklund A, Eken SM, Roy J, Eriksson P, Dacken B, Ramanujam D, Dueck A, Engelhardt S, Boon RA, Eckstein HH, Spin JM, Tsao PS, Maegdefessel L. H19 Induces Abdominal Aortic Aneurysm Development and Progression. Circulation. 2018;138(15):1551-1568.
- Kojima Y, Werner N, Ye J, Nanda V, Tsao N, Wang Y, Flores AM, Miller CL, Weissman I, Deng H, Xu B, Dalman RL, Eken SM, Pelisek J, Li Y, Maegdefessel L, Leeper NJ. Proefferocytic Therapy Promotes Transforming Growth Factor-β Signaling and Prevents Aneurysm Formation. 2018;137(7):750-753.
- Klaus V, Tanios-Schmies F, Reeps C, Trenner M, Geisbüsch S, Lohoefer F, Eckstein HH, Pelisek J. Cathepsin B and S are associated with the degraded product of collagen I in abdominal aortic aneurysm VASA. 2018;47(4):285-293.
- Klaus V, Tanios-Schmies F, Reeps C, Trenner M, Matevossian E, Eckstein HH, Pelisek J. Association of MMP levels with collagen degradation in the context of AAA. Eur J Vasc Endovasc Sur. 2017;53:549-558.
- Biehler J, Kehl S, Gee MW, Tanios F, Pelisek J, Maier A, Reeps C, Eckstein HH, Wall WA. Probabilistic Non-Invasive Prediction of Wall Properties of Abdominal Aortic Aneurysms Using Bayesian Regression Biomech Model Mechan. 2017;16(1):45-61.
- Gawinecka J, Schönrath F, von Eckardstein A. Acute aortic dissection: pathogenesis, risk factors and diagnosis. Swiss Med Wkly. 2017:w14489.
- Han Y, Tanios F, Reeps C, Zhang J, Schwamborn K, Lutz B, Eckstein HH, Zernecke A, Pelisek J. Histone acetylation and expression of corresponding histone acetyl-transferases in human abdominal aortic aneurysm. Clin Epigenetics. 2016; 8:3.
- Tanios F, Pelisek J, Lutz B, Reutersberg B, Kallmayer M, Matevossian E, Schwamborn K, Hösel V, Eckstein HH, Reeps C. CXCR4: a potential marker for inflammatory activity in abdominal aortic aneurysm wall. Eur J Vasc Endovasc Sur. 2015;50(6):745-753.
- Tanios F, Gee MW, Pelisek J, Kehl S, Biehler J, Haneder S, Grabher-Meier V, Wall WA, Eckstein HH. Reeps C. Interaction of biomechanics with extracellular matrix components in abdominal aortic aneurysm wall. Eur J Vasc Endovasc Surg. 2015;50(2):167-174.
- Lohoefer F, Reeps C, Lipp C, Rudelius M, Haertl M, Eckstein HH, Pelisek J. Quantitative expression and localisation of cysteine proteases in human abdominal aortic aneurysm. Exp Mol Med. 2014;9(6):e90846.
- Reeps C, Kehl S, Tanios F, Biehler J, Pelisek J, Maier A, Wall WA, Eckstein HH, Gee MW. Biomechanics and gene expression in abdominal aortic aneurysm. J Vasc Surg, 2014;60(6):1640-1647.e2.
- Reeps A, Maier A, Pelisek J, Härtl F, Grabher-Meier V, Wall WA, Essler M, Eckstein HH, Gee M. Measuring and Modeling the Patient-Specific Distributions of Material Properties in Abdominal Aortic Aneurysm Wall. Biomech Model Mechanobiol, 2013;12(4):717-733.
- Reeps C, Bundschuh RA, Pelisek J, Herz M, van Marwick S, Schweiger M, Eckstein HH, Nekolla SG, Essler M. (2013) Quantitative assessment of glucose metabolism in the vessel wall of abdominal aortic aneurysms: correlation with histology and role of partial volume correction. Int J Cardiovasc Imaging, 2013;29(2):505-512.
- Härtl F, Reeps C, Wilhelm M, Ockert S, Zimmermann A, Eckstein HH. Offene und endovaskuläre Therapie des abdominalen Aortenaneurysmas: Krankheitsbild – Evidenz – Ergebnisse. Dtsch Med Wochenschr 2012; 137(24): 1303-1308.
- Lipp C, Lohoefer F, Reeps C, Rudelius M, Baumann M, Heemann U, Eckstein HH, Pelisek J. Expression of a disintegrin and metalloprotease family of proteins in human abdominal aortic aneurysms. J Vasc Res. 2012;49(3):198-206.
- Lohoefer F, Reeps C, Lipp C, Rudelius M, Zimmermann A, Eckstein HH, Pelisek J. Histological analysis and localisation of cathepsins in human abdominal aortic aneurysm. Int J Exp Pathol, 2012;93(4):252-258.
- Reeps C, Pelisek J, Seidl S, Schuster T, Zimmermann A, Wendorff H, Kuehnl A, Eckstein HH. Inflammatory infiltrates and neovessels are relevant sources of MMPs in abdominal aortic aneurysm wall. Pathobiology. 2009;76(5):243-252.
- Reeps C, Gee MW, Maier A, Pelisek J, Gurdan M, Wall W, Mariss J, Eckstein HH, Essler M. Glucose Metabolism in the Vessel Wall Correlates With Mechanical Instability and Inflammatory Changes in a Patient With a Growing Aneurysm of the Abdominal Aorta. Circulation – Cardiovasc Imaging. 2009;2(6):507-509.