Research Group Alexander Ring
Keywords
Next-generation liquid biopsies, personalized immunotherapy, metastatic cancer treatment
Summary & Mission statement
Summary
Our work integrates cytapheresis-based ultra-high-yield liquid biopsy with next-generation sequencing and profiling of circulating tumor and immune cells, enabling the discovery of actionable targets and the design of comprehensive, personalized tumor vaccines for patients with metastatic cancer.
Mission statement
Our mission is to revolutionize precision oncology by leveraging next-generation liquid biopsy and cell profiling to capture the temporal and spatial heterogeneity of metastatic cancer, enabling comprehensive, real-time targeting of multifocal tumors and transforming outcomes for patients with otherwise treatment-resistant disease.
Overview
Introduction
While precision oncology has led to remarkable progress, many patients with metastatic cancers still lack effective, personalized treatment options. Tissue biopsies—the current gold standard—are invasive and prone to sampling bias, failing to capture the full heterogeneity of metastatic disease. To overcome these limitations, researchers at the University Hospital Zurich (USZ) and ETH Zurich have developed a novel cytapheresis-based liquid biopsy platform that enables minimally invasive, large-scale isolation of circulating tumor cells (CTCs) and immune cells from blood.
Achievements
Building on an established clinical-research infrastructure at USZ and ETH Zurich, the feasibility and safety of CTC apheresis have already been demonstrated in 25 patients. These early-phase procedures yielded high numbers of CTCs and immune cells, with successful downstream molecular processing, including sequencing and cell culture. These results validate the robustness of the approach and provide a strong technical and clinical foundation for expanded clinical application.
Aims
The current study aims to (1) increase patient enrollment for large-scale CTC and immune cell apheresis, (2) identify actionable mutations and immunogenic neoantigens in CTCs, and (3) functionally profile circulating immune cells. Ultimately, the project seeks to enable personalized immunotherapy strategies, including neoantigen-targeted tumor vaccines, for hard-to-treat metastatic cancers.
Publications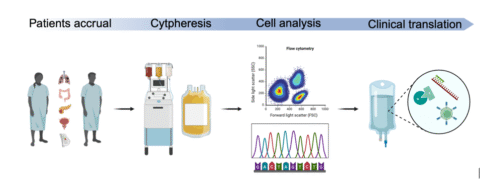
In a nutshell: Our goal is to build a robust clinical-translational diagnostic platform to conduct molecular and functional profiling of circulating tumor cells (CTCs) and tumor-associated immune cells harvested from metastatic cancer patients using next-generation large-scale liquid biopsies via cytapheresis. This technique collects cells at quantities 1,000 to 10,000 times higher than conventional methods, allowing for unprecedented comprehensive profiling through advanced next-generation sequencing (NGS), multi-parametric phenotyping, and bioinformatics analysis. We aim to identify novel, clinically relevant biomarkers, including actionable mutations, tumor-associated neoantigens, and immune cell abundance, phenotype, and functionality. These findings will help predict responses to targeted therapies and immunotherapy, providing critical information for next-generation personalized immunotherapies such as tumor vaccines.
