Devices & systems
- 2 container washers
- 10 Cleaning and disinfection devices
- 4 Sterilizers
- 1 plasma sterilizer

The University Hospital Zurich has a specialized reprocessing unit for medical devices (AEMP) at the USZ Logistics and Service Center in Schlieren.
Our AEMP supports an efficient and responsible supply of operating theaters and clinics with the professional reprocessing of medical products. Surgical instruments and reusable medical devices are cleaned, disinfected, sterilized and made available in the AEMP in accordance with the highest quality and hygiene standards.
Thanks to the advanced technical equipment and the expertise of our sterilization specialists, the AEMP is one of the most modern sterile processing facilities in Europe.
At the AEMP, we actively strive to further develop sustainable and future-oriented sterile supply and, as a training company, promote various training and further education courses. We offer apprenticeships as a medical device technologist (EFZ) and internships to obtain a certificate of technical competence. Sterilization assistant.
Are you interested in placing your medical instruments in professional hands for reprocessing? The USZ has also been offering cleaning, disinfection and sterilization services for third parties since 2021.
Contact nowThe AEMP at the USZ boasts a state-of-the-art infrastructure. Our devices and systems are among the most advanced in the whole of Europe – they are the basis for efficient sterile supply with consistently high process quality.
The core task of the AEMP is the reprocessing of medical products. We also guarantee seamless monitoring of the individual process stages and ensure smooth picking and delivery of the sterile goods.
The AEMP is an important part of the sterile goods cycle and thus contributes to a responsible basis for medical interventions.
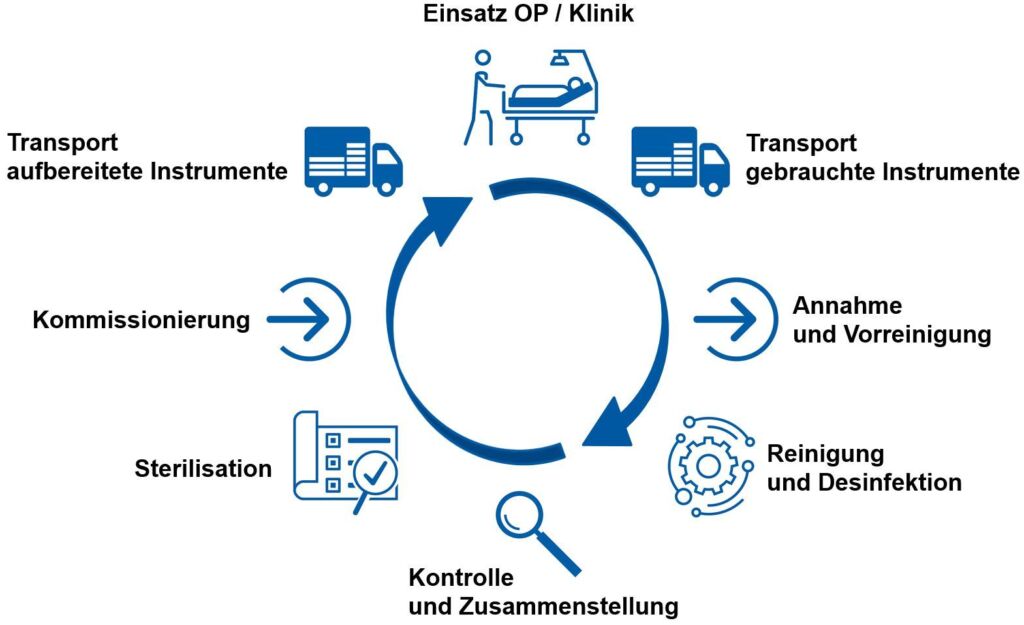
In the AEMP, medical instruments are reprocessed in accordance with the latest Swissmedic safety and hygiene standards and prepared for surgical procedures. Sterile processing consists of the following sub-processes:
The USZ offers GDP-compliant transportation of sterile goods in cooperation with an external transport service provider. The goods are packaged securely, delivered and collected reliably and on time according to a fixed schedule.
Express transportation is possible for orders at short notice.
The trays and instruments can be tracked, checked and organized at any time using an individual set code. End-to-end tracking is a prerequisite for optimum process reliability, predictability and needs-based supply of the OPS.
The accuracy and reliability of the AEMP are the result of strong teamwork. Our department is multicultural and diverse and offers a safe, respectful and carefree working environment for all employees. Good communication, mutual support and appreciation have the highest priority. The AEMP team works responsibly, efficiently and in a solution-oriented manner and is therefore extremely resilient even in exceptional situations.
Our department is divided into production, support, instrument management and quality management. These four organizational units are in close contact with each other and work together to drive the company forward. The AEMP continuously trains and develops employees. It is important to us to pass on specialist knowledge and expertise in the long term and to benefit from both our experience and the latest scientific findings.
ISO certificationThe AEMP of the University Hospital Zurich has an ISO-certified quality management system that ensures the highest quality and safety standards in the field of medical device technology. |
 |