Bachelor, Master and Doctoral Thesis
The group offers Bachelor, Master and Doctoral thesis with a clinically research focus. Candidates interested may contact us.
Our research aims at the identification and characterization of genes and pathways which may represent potential prognostic and predictive markers ('translational research') in human cancer. Genome and transcriptome analysis as well as the characterization of genetic alterations with their functional impacts in specific human tumors, primarily renal cell cancer, are of special interest.
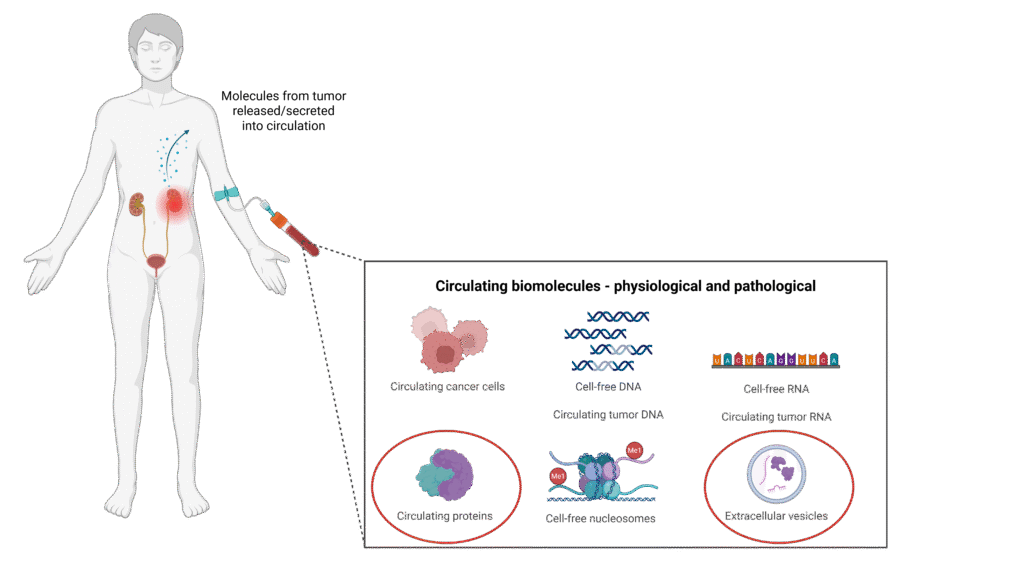
Clear cell renal cell carcinoma is the most prevalent kidney tumour, however it remains underserved by molecular biomarkers, marring precise patient management in clinics. Identifying biomarkers that can be detected using minimally invasive techniques can allow for the longitudinal monitoring of patients in clinics. Towards this end, we are identifying and validating different classes of biomolecules, including protein, extracellular vesicles and circulating tumour DNA as molecular biomarkers in patient blood samples.
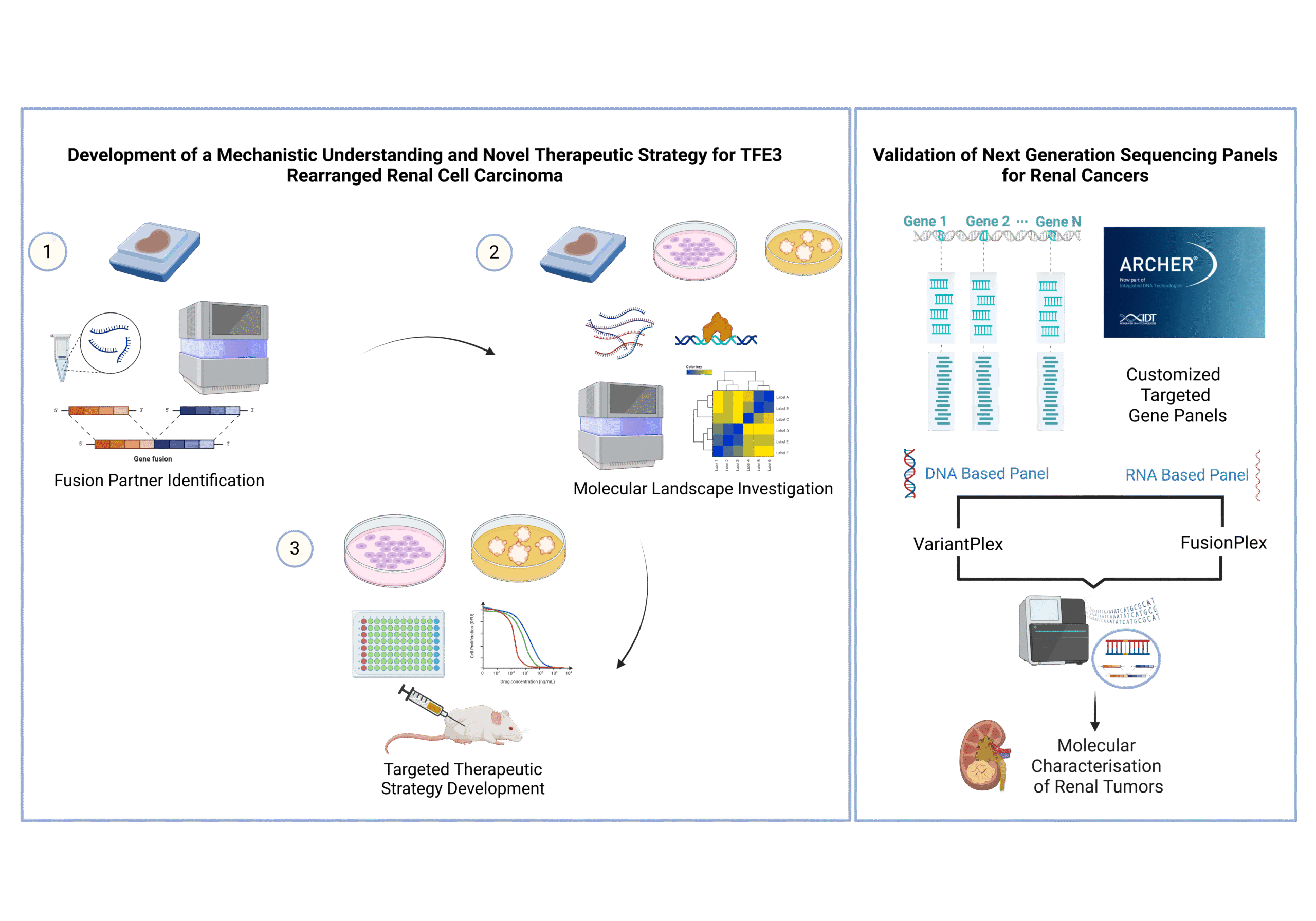
Molecular subtyping of renal tumours contributes to accurate diagnosis as well as provides a basis for personalized treatment. Large proportion of metastatic renal cell carcinomas remain under studied and for most of these entities, standard therapies are still lacking due to small patient numbers and limited knowledge about molecular profiles. In this study, we are working on customized targeted next generation sequencing panels in order to characterize different entities of renal tumours as well as unclassified cases. We further extend this project with a special focus on TFE3 rearranged renal cell carcinoma, where we want to investigate the molecular landscape and disease biology with the help of high throughput technologies. Finally, we aim to uncover specific vulnerabilities of TFE3-tRCC that prompt the development of new targeted therapeutic strategies.
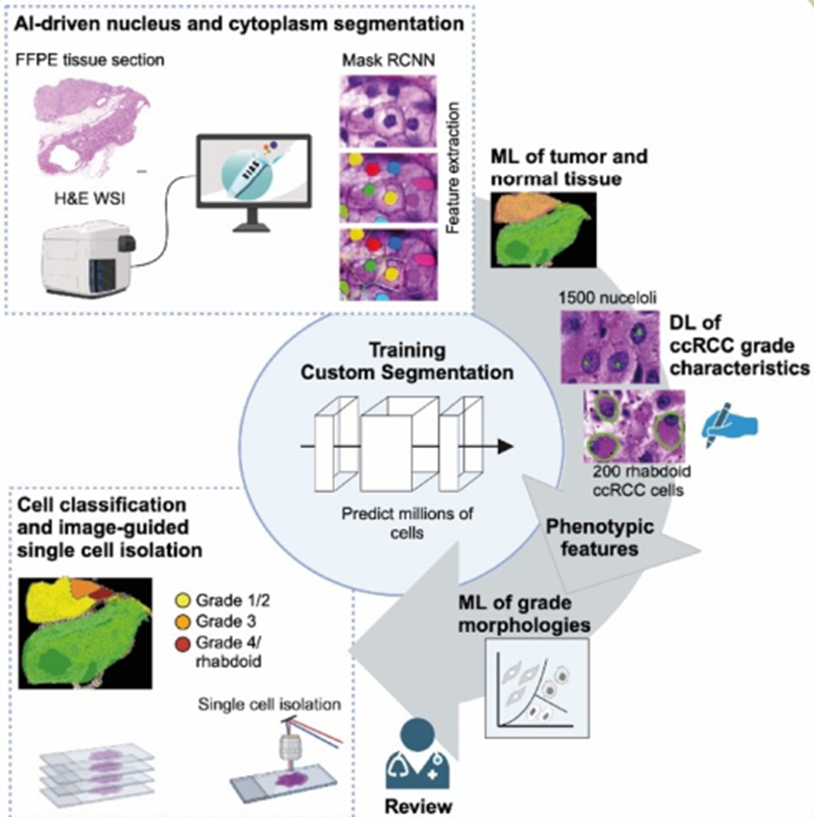
In this study, we integrated advanced AI-based image analysis with single-cell isolation and multi-omics profiling to dissect the link between clinically relevant morphological and molecular features of ccRCC cells. Using a novel digital pathology workflow, we precisely quantified low-grade, high-grade, and rhabdoid morphologies in ccRCC diagnostic images. By isolating two sets of morphologically distinct cells for detailed mRNA and protein expression analyses we uncovered increasing dysregulation of cancer-relevant molecular pathways associating with higher histopathological grades.
The group offers Bachelor, Master and Doctoral thesis with a clinically research focus. Candidates interested may contact us.