Research Group Manuel Stucki
Keywords
DNA repair, Genome instability, Chromatin, Breast and Ovarian cancer
Summary & Mission statement
The primary goal of our research is to understand how cells detect, signal and repair DNA damage in order to maintain the integrity of their genome. The larger objective is to explore how such insights can be translated towards more effective and better-tolerated anti-cancer therapies.
Overview
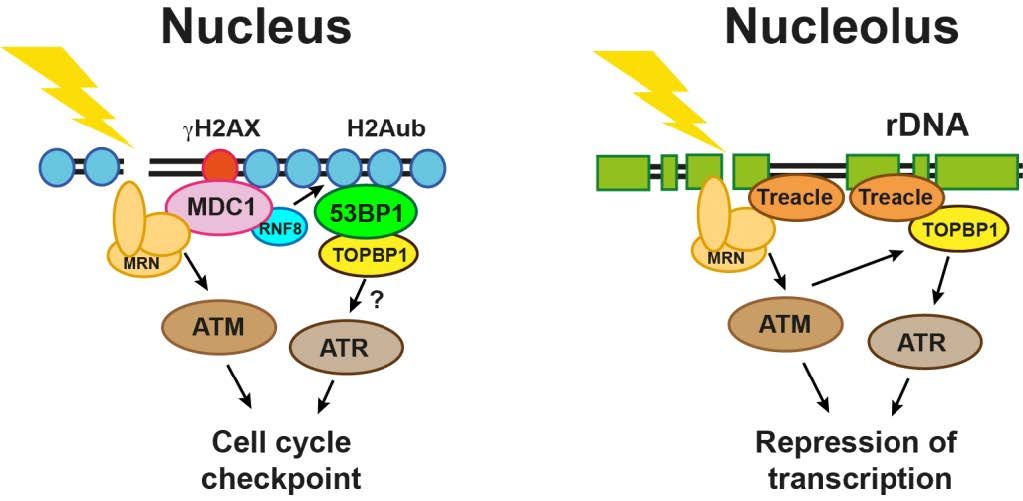
My lab has a long-standing interest in a group of proteins that are usually referred to as adaptors/mediators of the DNA damage response. Some of these factors act as tumor suppressors and are frequently mutated in hereditary breast and ovarian cancer (e.g. BRCA1). One of the major physiological roles of these proteins is to regulate DNA repair pathway choice during the cell cycle by orchestrating the dynamic localization of DNA damage response factors at sites of DNA lesions.
Currently, we are specifically focusing on the question how cells deal with DNA lesions in the highly compacted chromatin environment that exists during mitosis. In addition, we are also interested in how cells respond to DNA lesions in the nucleoli, small subnuclear compartments that accommodate the highly transcribed and repetitive rDNA.
To achieve our research goals, we combine biochemical and genetic approaches with advanced cell imaging technologies, including live cell imaging, software-assisted image analysis and super-resolution microscopy. We are also employing genome editing tools to generate loss-of-function or gain-of-function mutants in established human cell lines. In addition, we have access to fresh tumor tissue that allows us to validate our basic findings in clinically relevant samples.