Nat commun.
Engineering an anti-her2 biparatopic antibody with a multimodal mechanism of action
Kast F, Schwill M, Stüber JC, Pfundstein S, Nagy-Davidescu G, Rodríguez JMM, Seehusen F, Richter CP, Honegger A, Hartmann KP, Weber TG, Kroener F, Ernst P, Piehler J, Plückthun A. *CCCZ Members in bold
Abstract
The receptor tyrosine kinase HER2 acts as oncogenic driver in numerous cancers. Usually, the gene is amplified, resulting in receptor overexpression, massively increased signaling and unchecked proliferation. However, tumors become frequently addicted to oncogenes and hence are druggable by targeted interventions. Here, we design an anti-HER2 biparatopic and tetravalent IgG fusion with a multimodal mechanism of action. The molecule first induces HER2 clustering into inactive complexes, evidenced by reduced mobility of surface HER2. However, in contrast to our earlier binders based on DARPins, clusters of HER2 are thereafter robustly internalized and quantitatively degraded. This multimodal mechanism of action is found only in few of the tetravalent constructs investigated, which must target specific epitopes on HER2 in a defined geometric arrangement. The inhibitory effect of our antibody as single agent surpasses the combination of trastuzumab and pertuzumab as well as its parental mAbs in vitro and it is effective in a xenograft model.
Nat Commun. 2021 Jun 18;12(1):3790. doi: 10.1038/s41467-021-23948-6
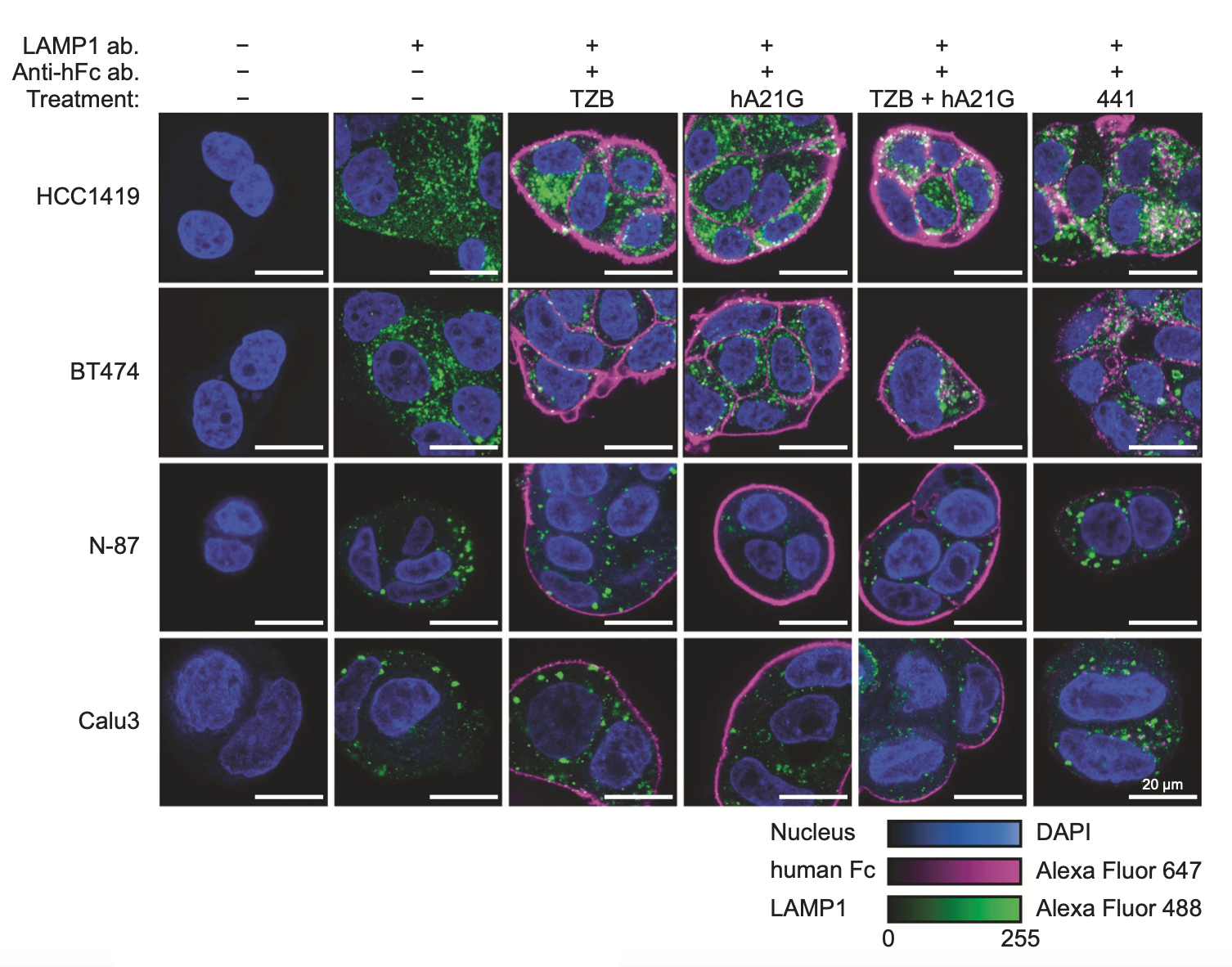
The engineered bi-paratopic antibody termed „441“ induces internalization and degradation in HER2-overexpressing cell lines, while all controls do not. This is seen by the disappearance of the continuous staining (magenta) on the outside, and the appearance of punctate staining in lysosomes (green). This effect is only seen with this particular specially engineered biparatopic construct, while other similar antibodies do not show this. Because of this unique effectivity in tumor xenografts, the antibody is now further developed for clinical trials.

