Sci Transl Med.
Schneider MA, Heeb L, Beffinger MM, Pantelyushin S, Linecker M, Roth L, Lehmann K, Ungethüm U, Kobold S, Graf R, van den Broek M, Vom Berg J, Gupta A, Clavien PA. *CCCZ Members in bold
Abstract
Platelet-derived peripheral serotonin has pleiotropic effects on coagulation, metabolism, tissue regeneration, and cancer growth; however, the effect of serotonin on the tumor microenvironment remains understudied. Peripheral serotonin–deficient (Tph1−/−) mice displayed reduced growth of subcutaneous and orthotopically injected syngeneic murine pancreatic and colorectal cancers with enhanced accumulation of functional CD8+ T cells compared to control C57BL/6 mice, resulting in extended overall survival. Subcutaneous and orthotopic syngeneic tumors from Tph1−/− mice expressed less programmed cell death 1 ligand 1 (PD-L1), suggesting serotonin-mediated regulation. Serotonin enhanced expression of PD-L1 on mouse and human cancer cells in vitro via serotonylation, which is the formation of covalent bonds between glutamine residues and serotonin, resulting in activation of small G proteins. Serotonin concentrations in metastases of patients with abdominal tumors negatively correlated to the number of CD8+ tumor-infiltrating T cells. Depletion of serotonin cargo or inhibition of serotonin release from thrombocytes decreased growth of syngeneic pancreatic and colorectal tumors in wild-type mice, increased CD8+ T cell influx, and decreased PD-L1 expression. Pharmacological serotonin depletion with oral fluoxetine or intraperitoneal injection of the TPH1 inhibitor telotristat augmented the effects of programmed cell death protein 1 (PD-1) checkpoint blockade and triggered long-term tumor control in mice subcutaneously inoculated with syngeneic colorectal and pancreatic tumors. Overall, peripheral serotonin weakens effector functions of CD8+ T cells within tumors. Clinically approved serotonin targeting agents alone or in combination with PD-1 blockade provided long-term control of established tumors in murine models, warranting further investigation of the clinical translatability of these findings.
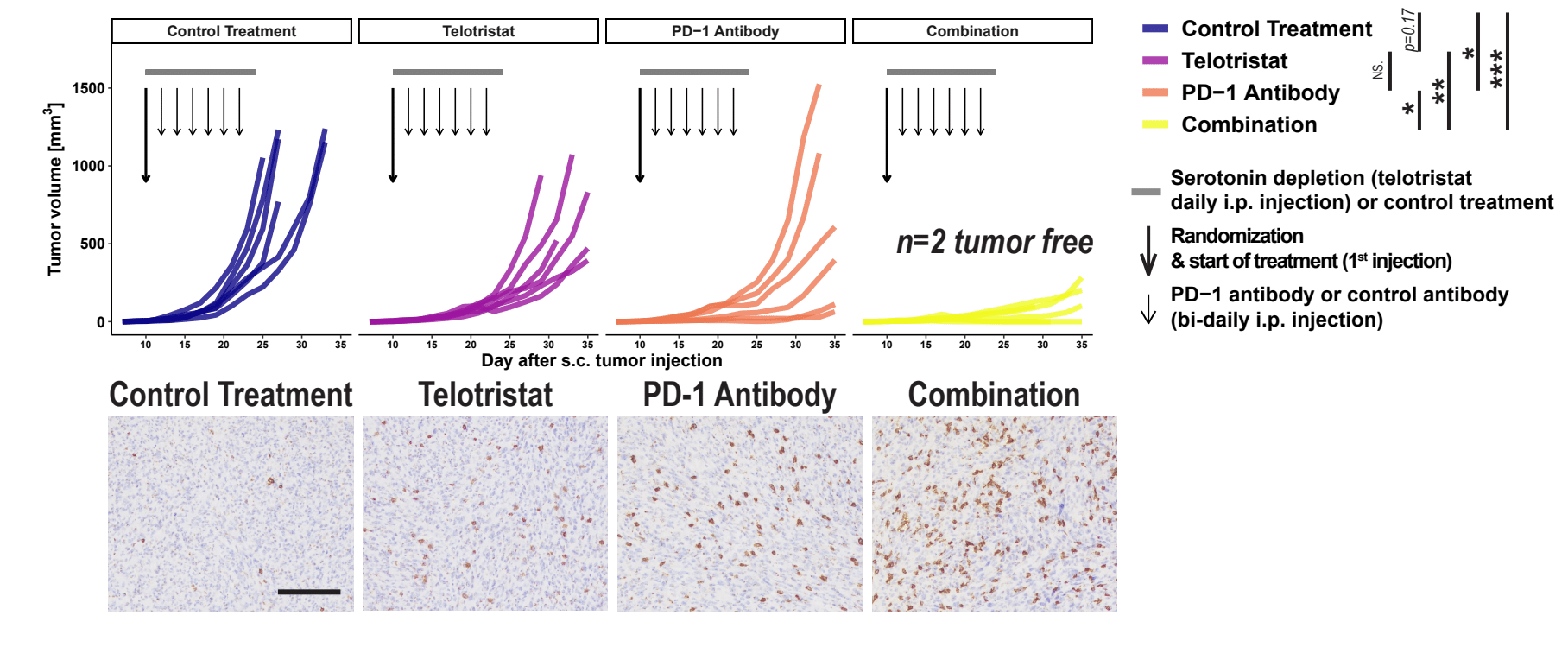
Tumor growth and immunohistochemical images of CD8a+ T cells in tumors of C57BL/6J mice after subcutaneous injection of 5 × 105 syngeneic MC38 cells randomized on day 10 and treated with either control injections, the specific Tph1 inhibitor telotristat, PD-1 antibody, or combination of telotristat plus PD-1 antibody. The bold black arrow indicates randomization and start of treatment, narrow black arrows show time points of PD-1 antibody or control treatments, and the gray horizontal bar shows duration of telotristat or control treatment. Specific serotonin depletion by daily telotristat injections reduced tumor growth comparable to bidaily anti-PD-1 monotherapy. Combination of telotristat and anti-PD-1 immunotherapy led to greater tumor control than either treatment alone with an eradication rate of 33% and increased numbers of infiltrating CD8+ T cells in tumors.
Sci Transl Med. 2021 Sep 15;13(611):eabc8188
doi: 10.1126/scitranslmed.abc8188

